Introduction
Mutations in the Janus Kinase 2 gene (JAK2) are commonly encountered in patients with myeloproliferative neoplasms (MPN) including polycythemia vera (PV) and essential thrombocythemia. JAK2 mutations particularly V617F is associated with PV in 98% cases. Gain of function mutations in the JAK2 exon 12 are reported to be associated with isolated erythropoiesis. Atypical mutations in JAK2 including (c.3323A>G, p.N1108S) were reported in patients with MPN who tested negative for the established driver mutations including exon 14 JAK 2, exon 9 CALR and exon 10 MPL genes. Primary familial congenital polycythemia (PFCP) is a rare disease inherited in an autosomal dominant fashion caused by hypersensitivity of EPO receptor. In such cases, an EPO receptor mutation is only found in 12-15% of affected patients.
Due to associated increased risk of thrombosis, bleeding, leukemic involvement and myelofibrosis in PV, bone marrow evaluation is essential in this patient population. In patients with polycythemia, the goals of treatment are to maintain appropriate level of hemoglobin/hematocrit and to reduce severity of symptoms.
We present a family affected by polycythemia and leukocytosis associated with an atypical heterozygous variant of JAK2 [c.3323A>G (p.Asn1108Ser)] currently reported as benign or variant of unknown significance (VUS) in literature.
Methods
Case series, retrospective electronic medical record review with institutional board review exemption
Results
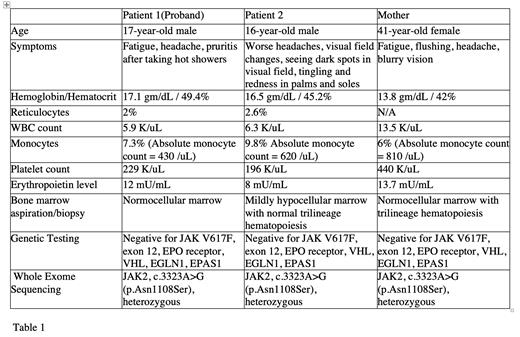
We report a case series of three Caucasian family members affected with JAK2 VUS mutation (mother and two sons). The proband is a 17-year-old male who presented to an outside facility at the age of 14 with complaints of fatigue, headache, and intense pruritis after hot showers. Initial workup was significant for marked erythrocytosis and reticulocytosis without evidence of abnormal cells on peripheral film. Bone marrow evaluation showed trilineage hematopoiesis with no morphological features of a myeloproliferative neoplasm. Genetic testing was negative for JAK2 V617F and exon 12 mutations. However, whole exome sequencing (WES) revealed a heterozygous mutation of JAK2 gene, c.3323A>G (p.Asn1108Ser). Additional investigation on proband's younger brother, who complained of similar symptoms, also revealed the same JAK2 variant. The proband's mother reported non-specific symptoms including headache, flushing, and occasional blurry vision that she previously attributed to hypertension. Her labs were significant for leukocytosis, monocytosis and thrombocytosis in the presence of normal red blood cell count and WES showed the same mutation. Bone marrow evaluation of the brother and the mother showed normal trilineage hematopoiesis and was negative for malignancy (Table 1). Proband's father is reportedly negative for the aforesaid mutation and is clinically asymptomatic.
The proband and his brother are currently managed with phlebotomy every 3 to 4 weeks to maintain hematocrit below 45%, leading to improvement in symptoms. Cytoreductive therapies have been discussed but not initiated. Proband's mother is managed by an adult hematologist. Scheduled phlebotomies with close surveillance is recommended for clinical practice along with interval research sequencing.
Conclusion
Our case series reports a family with clinical features and laboratories suggestive of a familial form of polycythemia, WES detected the presence of an atypical JAK2 mutation: c.3323A>G, (p.Asn1108Ser). The phenotypical and clinical significance of this mutation has been considered non canonical per reports and classified as likely benign by reputable commercial genetic testing laboratories. However, other reports identify this JAK2 variant as having a significant increased prevalence in patients with MPNs and AML compared to the general population. It may result in a gain of function of the JAK2 protein and subsequent development of MPNs. Enrollment in research sequencing protocols is encouraged in such patients to closely follow the pathologic potential and spectrum of genetic variants rarely reported in literature. Additional research to determine the pathogenicity of this genetic variant is highly warranted.
Disclosures
DiNardo:Astellas: Honoraria; ImmuniOnc: Honoraria; Schrödinger: Consultancy; AbbVie/Genentech: Honoraria; Takeda: Honoraria; Novartis: Honoraria; BMS: Honoraria; Fogham: Honoraria; Notable Labs: Honoraria; Servier: Honoraria. Masarova:MorphoSys US: Membership on an entity's Board of Directors or advisory committees. Rodriguez:Sanofi: Membership on an entity's Board of Directors or advisory committees.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal